Michael Molitch-Hou from 3D Printing Industry

Organovo is steadily on track with its organ bioprinting research. Having conducted a number of studies, the company has already released its first cellular assay of liver tissue, exVive3D, for pharmaceutical testing purposes. Now, the bioprinting firm is moving onto human kidney tissue. At the 2015 Experimental Biology conference in Boston, today, Organovo gave attendees a look at the work done so far on their in vitro three-dimensional kidney tissue.
At the conference, Organovo presented their latest research, 3D printed a part of the duct system attached to the kidney, “kidney proximal tubular tissues“, using multiple cell types. Moreover, the tissue was able to survive in vitro for two weeks. This specific portion of the kidney may aid in the testing of medicines and the fact that they are made up of three different cell types will contribute to that application further, as well as provide a stepping stone to even more complex tissues.
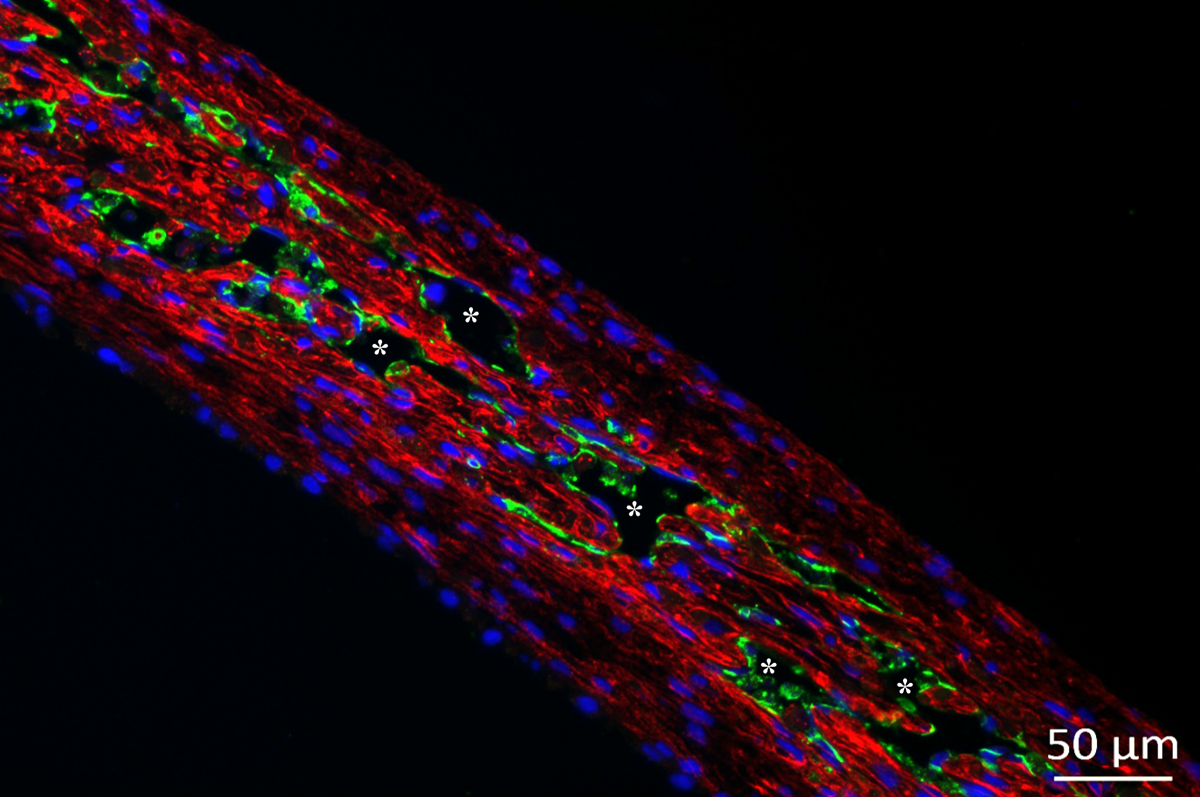
Credit: Organovo. “Organovo has described the first 3D bioprinted human kidney tissue, which is a significant advance over simple single layer cell cultures that are used today for drug testing and disease research. This image shows that the tissue is composed of different types of kidney cells that make up the proximal tubule, which plays a pivotal role in kidney disease. The tissue is 100% cellular (no scaffolds) with clear cellular connections and tissue architecture. (Not shown) The tissue also expresses normal kidney enzymes such as CYP450 (enzymes responsible for breaking down drugs) and gamma glutamyl transferase (GGT) activity, both of which are indicators of specific kidney cell (RPTEC) function. Key to colors and marks Tissues were stained with antibodies against two of the three different cell types found in the tissue: endothelial cell specific marker CD31 (green) and fibroblast marker TE7 (red). The third cell type is the polarized renal proximal tubular epithelial cells (RPTEC). The endothelial cells will associate with each other, forming networks. Over time, the center of these networks will “hollow out” to form microvessel lumens lined with epithelial cells. Networks with putative lumens lined with endothelial cells are marked (*).”
Organovo CTO and Executive Vice President of Research and Development, Dr. Sharon Presnell, explains, “Our bioprinted human kidney tissue represents a significant technical advance over the simple monolayer cell line cultures that predominate today. The histologic and functional features of the initial prototypes are compelling, and the in vitro durability of the system will likely enable the assessment of drug effects at chronic, physiologically relevant doses. Furthermore, the cellular complexity of the system will likely support mechanistic investigations into drug responses, including end points that have been difficult or impossible to assess in vitro, including tubular fibrosis and post-injury recovery.”
Kidney disease affects more than 25 million people in the United States and these printed tissues are a first step towards treating such diseases as polycystic kidney disease and, one day in the future, creating printed kidneys for transplant. Though that day is likely a long way off, printed organs would bypass the need for immunosuppressants that both prevent the body from rejecting alien organs and cause many post-transplant complications. Because that day is a long way off, however, organ donation is still vital to the survival of the more than 123,000 people on organ waiting lists in the US. This also means that the kidney tissue from Organovo will be used for pharmaceutical testing in the short term.
The company’s CEO, Keith Murphy, elaborates, “Kidney represents an ideal extension of Organovo’s capabilities to 3D bioprint organ tissues that can be tremendously useful in pharmaceutical research. The results released today admirably demonstrate a proof of concept that kidney is on the way to becoming another core commercial tissue for Organovo. The product that we intend to build from these initial results can be an excellent expansion for our core customers in toxicology, who regularly express to us an interest in having better solutions for the assessment of human kidney toxicity.”
Organovo states: “Nearly one in five drug candidates fail Phase 3 clinical trials because of kidney toxicity. Only a handful of poor models are used today to test a drug’s impact on the kidneys.” If the company can produce a new product, similar to that of their exVive3D liver assay, for the kidney, researchers may have a more viable method for testing drug toxicity on the kidneys, ensuring that medicines can get to market more quickly, with less of a chance of them being later removed from shelves, due to inadequate tools for proper research.
The tissue construct consists of three types of human cells: epithelial cells (RPTEC), a living interstitial layer made from renal fibroblasts (RF), and endothelial cells (EC), all of which have maintained their proper shape and survived for two weeks in vitro. Not only that, but microvascular structures grew in the tissue during this time. Or, as the company puts it:
The kidney proximal tubule tissue is multi-cellular and fully human, consisting of polarized renal proximal tubular epithelial cells (RPTEC) and a living interstitial layer comprised of renal fibroblasts (RF), and endothelial cells (EC). Immunohistochemical analysis shows clear evidence of intercellular junction formation (E-Cadherin+) between the epithelial cells, extensive formation of microvascular structures, and stable maintenance of the layered architecture for at least two weeks in vitro. The epithelial cells within the multi-layered tubular model express CYP450 mRNAs and possess gamma glutamyl transferase (GGT) activity, both of which are indicators of RPTEC function. Importantly, our initial characterization shows that the expression of GGT increases over two weeks post-fabrication, indicating a gain-of-function over time.
Image via onlinehumananatomycourse.net
Speaking with Loyola University Medical Center’s Dr. Susan Hou, a well-regarded nephrologist who is also my mom, I was able to gain a better understanding of what this specific portion of the kidney is: “The kidney is made up of filtering units called glomerulae; they’re collections of capillaries that filter the blood, the function of the kidney. They filtered probably 100x what you need to get rid of. And there are tubules made up of four parts: the proximal, the loop of Henle, distal, and the collecting duct. These take back all of the waste that you don’t need to get rid of. So, it’s great to make proximal tubular cells, but, without the glomerulus, you can’t have any function of the kidney.”
She wasn’t able to comment specifically on the exhibited function of the epithelial cells demonstrated by Organovo’s research, as I was reading the company’s press release to her over the phone and probably mispronouncing “gamma glutamyl transferase”, but she did underscore the importance of the fact that they were able to print the “living interstitial layer comprised of renal fibroblasts (RF), and endothelial cells (EC)”, saying, “They’re a little bit closer to making the whole kidney if they’ve got the proximal tubule in the right position with the part of the kidney that’s between the filtering units.” In other words, Organovo is making significant progress, but still has some ways to go before 3D printing an entire, functioning kidney.
